Intraoperative continuous noninvasive hemoglobin monitoring in patients with placenta previa undergoing cesarean section: a prospective observational study
Article information
Abstract
Background:
Obstetric patients with placenta previa are at risk for sever peripartum hemorrhage. Early detection of anemia and proper transfusion strategy are important for the management of obstetric hemorrhage. In this study, we assessed the utility and accuracy of noninvasive hemoglobin (SpHb) monitoring in patients with placenta previa during cesarean section.
Methods:
Parturients diagnosed with placenta previa and scheduled for cesarean section under spinal anesthesia were enrolled. SpHb and laboratory Hb (Lab-Hb) were measured during surgery as primary outcomes.
Results:
Seventy-four pairs of SpHb and Lab-Hb were collected from 39 patients. The correlation coefficient was 0.877 between SpHb and Lab-Hb (P < 0.001). The Bland–Altman plot showed a mean difference ± SD of 0.3 ± 0.8 g/dl between noninvasive Hb and Lab-Hb, and the limits of agreement were −1.2 to 1.8 g/dl. The magnitude of the difference between SpHb and Lab-Hb was < 0.5 g/dl in 64.9%; however, it was > 1.5 g/dl in 10.8%.
Conclusions:
SpHb monitoring had a good correlation with Lab-Hb. A small mean difference between SpHb and lab-Hb might not be clinically significant; however, the limits of agreements were not narrow. In particular, SpHb could be overestimated in the anemic population. Based on our results, further studies investigating the accuracy and precision of SpHb monitoring should be performed in parturients presenting Hb below 10 g/dl.
INTRODUCTION
Pregnant women present physiologic anemia during pregnancy, thus peripartum bleeding can cause more severe anemia during vaginal delivery or the cesarean section (C-sec). When parturients with placenta previa undergo C-sec, there may be increased chance of severe intraoperative bleeding, followed by profound anemia requiring massive blood transfusion. For the purpose of frequent laboratory evaluation, proper transfusion strategy, and vital sign monitoring, intraoperative invasive arterial monitoring is usually performed.
Noninvasive hemoglobin (Hb) monitoring is useful when requiring continuous monitoring of Hb and performing proper transfusion; it is applicable in various clinical situations, such as managing critically ill patients with bleeding tendency, trauma patients with continuous bleeding, as well as small pediatric patients with difficulty in frequent blood sampling, orthopedic operation with intraoperative substantial blood loss, or hemorrhagic shock [1–5]. However, this view remains contentious; some believe that noninvasive Hb monitoring may not be as accurate as the laboratory Hb, and thus asserting that it should not be used exclusively to guide transfusion [2,6,7].
During the peripartum period, it was found that noninvasive Hb monitoring was well correlated with laboratory Hb [8,9]. However, to the best of our knowledge, there is no study evaluating the usefulness of noninvasive Hb monitoring in parturient with placenta previa. When surgical bleeding occurs in parturients with placenta previa during C-sec, it is generally acute and extensively massive; therefore, transfusion is performed based on clinical implication until laboratory Hb results are obtained in such cases. If intraoperative Hb is monitored promptly and accurately by continuous noninvasive Hb monitoring in parturients with placenta previa, anemia can be detected instantly, which will allow proper fluid and transfusion management.
In this study, we applied continuous noninvasive Hb monitoring in parturients with placenta previa undergoing C-sec and compared the Hb level between continuous noninvasive Hb monitor and laboratory Hb level.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of Seoul National University Bundang Hospital (no. B-1706/403-306) and registered at ClinicalTrial.com (NCT 03409822).
Parturient women who underwent elective C-sec due to placenta previa were enrolled in this study. The exclusion criteria were as follows: American Society of Anesthesiology physical status 3 or more; refusal to participate in this study; peripheral vascular disease; cyanotic condition due to the congenital or acquired heart disease; refusal of transfusion; and inability to communicate in Korean. All participants provided written informed consent.
On arrival to the operating room, routine monitoring— such as electrocardiography, noninvasive blood pressure, and pulse oximetry—was initiated. In addition, invasive arterial pressure monitoring was also performed, and laboratory Hb (Lab-Hb) was evaluated using arterial blood via Stat Profile pHOx Ultra (Nova Biomedical, USA). Noninvasive Hb (SpHb) was measured by using the Radical-7 Pulse CO-oximeter (Masimo Corp., USA). Spinal anesthesia was performed using 7–9 mg of hyperbaric bupivacaine with 15 μg of fentanyl at lateral decubitus position, during which, 500 ml of colloid was routinely infused to prevent hypotension. Intraoperative hypotension was managed with phenylephrine or ephedrine, and bradycardia was treated with atropine. Spinal anesthesia was converted to general anesthesia if the operation was prolonged unexpectedly. Red blood cells (RBC) transfusion was initiated when Lab-Hb was below 8 g/dl, and the unit of RBC transfusion was decided according to the severity of surgical bleeding. SpHb was calibrated based on the initial lab-Hb before starting the operation. When the likelihood of massive and continued intraoperative bleeding was relatively high, evaluation of Lab-Hb and SpHb was at the discretion of the attending anesthesiologist.
The primary outcomes were SpHb and Lab-Hb. The secondary outcomes were perfusion index (Pi) measured simultaneously with SpHb by Radical-7 Pulse CO-oximeter, infused fluid volume, unit of transfusion, calculated blood loss during surgery, and urine output.
Statistical analysis
In previous studies [1,2,10,11], the range of correlation coefficient between SpHb and Lab-Hb was reported from 0.47 to 0.72. It was assumed that the correlation coefficient would be approximately 0.7 in our study. Adjusting for a 10% of dropout, 39 patients were required to give a statistical power of 80% and a type 1 error of 5%.
The characteristics of patients and clinical factors were presented as the mean and standard deviation (SD) or number (%). Correlation between SpHb and Lab-Hb was examined by Spearman’s rank correlation analysis. The differences between the two parameters were analyzed by one sample t-test. Bland–Altman analysis was used to assess the agreement between the two methods measuring Hb, with the limits of agreement defined as 2 SD from the mean. All statistical analyses were performed using IBM SPSS Statistics version 25.0 (IBM Corp., USA). P value of less than 0.05 was considered statistically significant.
RESULTS
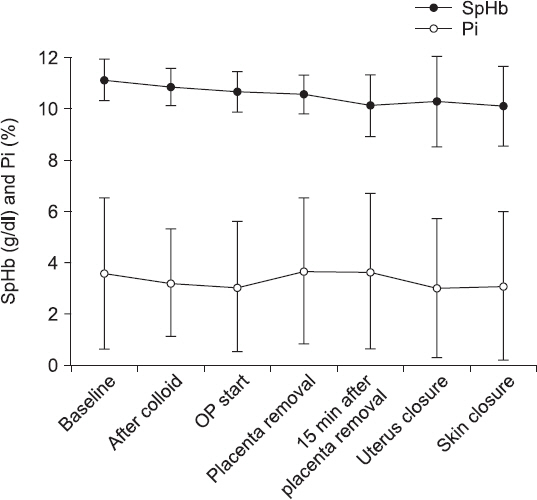
The characteristics of 39 patients, surgery, and anesthesia were shown in Table 1. The mean amount of blood transfusion and incidence of patients receiving transfusion during surgery were presented in Table 2. The change of SpHb and Pi were presented at each time point during anesthesia (Fig.1) After colloid loading during spinal anesthesia, the mean SpHb decreased by 0.3 g/dl.
A total of 74 pairs of SpHb and Lab-Hb were collected from 39 patients. The correlation coefficient between SpHb and Lab-Hb was 0.877 (P < 0.001; Fig. 2). The Bland–Altman plot showed a mean difference ± SD of 0.3 ± 0.8 g/dl between noninvasive Hb and Lab-Hb, and the limits of agreement ranged from −1.2 to 1.8 g/dl (Fig. 3).

Correlation between the laboratory hemoglobin (Lab-Hb) and noninvasive hemoglobin (SpHb). The correlation coefficient was 0.877 between the SpHb and Lab-Hb.
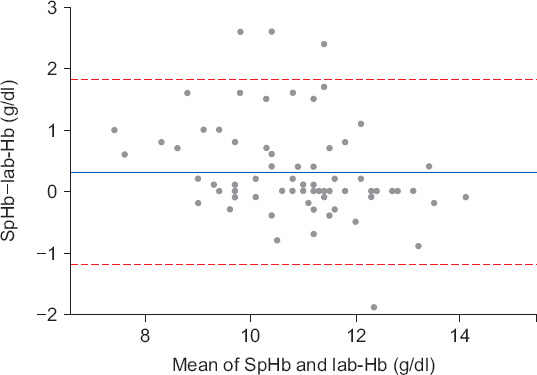
Bland–Altman analysis for laboratory hemoglobin (Lab-Hb) and noninvasive hemoglobin (SpHb). The mean difference (one horizontal solid line) between the SpHb and Lab-Hb was 0.3 g/dl and the limits of agreement (two horizontal dash lines) were −1.2 to 1.8 g/dl.
The magnitude of difference between SpHb and Lab-Hb was < 0.5 g/dl in 64.9%; however, its difference was 1.5 g/dl in 10.8% (Table 3). When the subgroups were classified according to Pi values, there were significantly more patients in the Pi ≥ 2.0 subgroup presenting low discrepancy between SpHb and Lab-Hb (P = 0.042).
DISCUSSION
The aim of this study was to determine the agreement of SpHb against Lab-Hb in C-sec causing massive and acute intraoperative blood loss, and our findings showed that SpHb had a good correlation with Lab-Hb.
In practice, a laboratory Hb is usually considered as the gold standard for measuring intraoperative Hb. However, laboratory Hb measurements only provide intermittent and delayed results in the clinical condition of massive and acute blood loss due primarily to its turnaround time. Therefore, frequent laboratory evaluation is required to detect the decrease of Hb level during an operation, which is not only time-consuming but also labor intensive, requiring specific trained personnel.
On the other hands, continuous noninvasive Hb monitoring can obtain real-time change of Hb. The accuracy of noninvasive Hb monitoring has been reported in several surgeries presenting significant intraoperative bleeding [12–14]. In parturient undergoing C-sec, noninvasive Hb was also evaluated at preand post-operative periods and it did not seem to be as accurate as the laboratory one [15,16]. Different from the previous studies, this study enrolled the pregnant population with placenta previa; therefore, intraoperative massive and rapid blood loss was expected. In addition, we focused the correlation between the two variables only during the intraoperative period, when substantial blood loss is expected to occur and immediate transfusion can be initiated.
In our results, the correlation coefficient between the two measurements was relatively high; therefore, SpHb may be valuable in continuous monitoring Hb trend. However, a high correlation coefficient does not imply that there is a good agreement between the two methods. Thus, the focus should be more on the results of Bland–Altman analysis. On average, SpHb was recorded to be 0.3 g/dl higher than LabHb according to the Bland–Altman plot. This small difference did not seem to be a problem in clinical setting, especially in situations where transfusion is appropriate. However, the limits of agreements were not narrow enough to be acceptable for clinical purpose, such as detection of anemia triggering RBC transfusion. In addition, we found a slight negative trend of the differences, proportional to the increment of the measurement. When we regressed the differences of the two Hb measurements on their average, the standardized coefficient for the mean differences was −0.3. Although the coefficient was very small and only 15 datasets existed in the mean Hb level of < 10 g/dl, it is important to keep in mind that SpHb could be measured higher than the Lab-Hb in anemic conditions. Even though SpHb and Lab-Hb were calibrated initially with each other, there could be a deviation between SpHb and Lab-Hb in anemic conditions after substantial blood loss. If SpHb was overestimated in the anemic population, a decision to initiate RBC transfusion could be delayed. It was reported that adding SpHb to the standard laboratory Hb measurement decreased the volume of transfusion in surgeries with high blood loss, such as several neurosurgeries [17], and SpHb was remarkably consistent with Lab-Hb in trauma patients with Hb under 8 g/dl [18]. However, it remains controversial whether SpHb is enough to detect severe anemia or to suggest the need for RBC transfusion solely [10,19,20].
In our patients, Pi measured simultaneously with SpHb did not change drastically during surgery; although, there were individual differences. Pi should be considered to evaluate the accuracy of SpHb, and it has been reported that SpHb is more accurate in patients with Pi > 2.0 [21–23]. In our study, SpHb also tended to be more consistent with Lab-Hb in the subgroup pf Pi ≥ 2.0. Thus, interpretation of SpHb should be made with caution in clinical conditions with decreasing Pi, such as hypothermia, vasoactive drugs, or increased sympathetic tone.
When we use continuous and noninvasive Hb monitoring, it should be confirmed whether it is really helpful in the transfusion practice, such as timely initiation of transfusion, avoidance of over-transfusion, and proper amount of transfusion. In the present study, transfusion was performed based on Lab-Hb; thus, we could not reveal the effectiveness of SpHb on the transfusion practice. Further study is necessary to evaluate this issue.
According to the previous report, Hb of venous blood was higher than that of arterial blood [24]. In addition, Hb measured by co-oximetry method was shown to be higher than Hb measured in central laboratory (reported as complete blood counts) [25]. In our study, Lab-Hb was measured using arterial blood via the co-oximetry method. Depending on which method is set as the gold standard of Hb measurement, there can be larger or smaller differences between SpHb and Lab-Hb. It should be considered carefully that a larger bias is likely to cause concern in clinical diagnosis of anemia triggering RBC transfusion.
This study had several limitations. First, the datasets of SpHb and Lab-Hb in anemic conditions, especially 8 g/dl or less, were definitely lacking. When anemia was noticed and massive bleeding continued, RBC transfusion was initiated instantly to our participants. Therefore, severe anemic condition was rare in this study. Second, we did not consider the effects of vasoconstrictors used during the surgery. After spinal anesthesia, several parturients presented hypotensive events, when it was treated with phenylephrine or ephedrine with fluid administration. In addition, spinal anesthesia was converted to general anesthesia in two patients during the surgery. Thus, dynamic change probably occurred. As a result, the range of observed perfusion index on noninvasive Hb monitoring was from 0.47 to 9.2 in this study.
In conclusion, our study showed that SpHb monitoring had a good correlation with Lab-Hb. A small mean difference between noninvasive SpHb and Lab-Hb might not be clinically significant; however, the limits of agreements was not narrow. In particular, SpHb could be overestimated in the anemic population. Based on our results, further studies investigating the accuracy and precision of noninvasive Hb monitoring should be performed in parturients presenting Hb below 10 g/dl.
ACKNOWLEDGMENTS
This research was supported by the 2017 Research Grant funded by a Korean Society of Obstetric Anesthesiologists.
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
ORCID
Hyunsung Kim: https://orcid.org/0000-0001-7681-9280
Sang-Hwan Do: https://orcid.org/0000-0001-5452-4166
Jung-Won Hwang: https://orcid.org/0000-0002-0887-6889