INTRODUCTION
Anaphylaxis is a rapid-onset systemic hypersensitivity reaction that develops when an individual previously sensitized to a specific antigen is re-exposed to the same antigen. Activated basophils and mast cells release histamine and protease (tryptase) in a type I hypersensitivity reaction and then synthesize strong inflammatory substances such as leukotriene, prostaglandin and platelet activating factors via phospholipid metabolism [
1]. Anaphylaxis during the perioperative period is rare, with rates of 1 : 2,800 to 1 : 20,000 [
2-
4], but it can cause severe cardiovascular and respiratory collapse that can be lethal and comprises up to 3% of anesthesia-related deaths [
2-
5].
ăWhen these types of hypersensitivity reaction are suspected, the intradermal skin test is the most commonly used diagnostic tool for identifying the cause [
3]. Particularly, intradermal skin test should be confirmed as negative before using the antibiotics because of the high risk of anaphylaxis due to them. We report a case of anaphylactic shock occurring during general anesthesia in response to an intradermal skin test-negative antibiotic in a patient who had previously been under general anesthesia five times.
CASE REPORT
A 42-year-old female, height 175 cm and weight 52 kg, was admitted for resection of an intra-abdominal leiomyosarcoma. Thiopental sodium and vecuronium had been used in two of five previous operations consisting of two caesarean sections, a myomectomy, a transabdominal hysterectomy, and the excision of an intra-abdominal leiomyosarcoma, and propofol and rocuronium had been used in the other three operations. There was no known history of surgery- or anesthesia-related complications, asthma, atopy, or allergies. Preoperative evaluations including chest radiography, electrocardiography, and blood laboratory tests were normal.
As preoperative treatment, intramuscular atropine 0.5 mg and midazolam 2.0 mg were given 30 minutes before the operation. After her arrival in the operating room, the patientâs vital signs were monitored by noninvasive blood pressure measurement, electrocardiography, and pulse oximetry, and her mental status was evaluated by bispectral index (BIS) monitoring. Propofol and rocuronium were injected intravenously to induce anesthesia and after intubation, O
2-air-desflurane was used for maintenance of anesthesia. The preoperative intradermal skin test-negative antibiotic, flomoxef sodium, was injected intravenously just before the operation. During surgery, the patientâs status was confirmed by monitoring systolic/diastolic pressure 110-130/70-80 mmHg, pulse rate 80-100 beats/min, oxygen saturation 99%, and BIS 35-55. Twenty-five minutes after the operation, oxygen saturation dropped abruptly to 86%. Simultaneously, peak airway pressure (PAP) increased from 19 cmH
2O to 33 cmH
2O, and a wheezing sound was auscultated in both lung fields. Arterial blood gas analysis (ABGA) showed pH 7.32, pCO
2 44 mmHg, pO
2 55 mmHg, and Base excess 0.5 mM at a fraction of inspired oxygen (FiO
2) 0.3. After increasing FiO
2 to 1.0, ventolin was sprayed via an endotracheal tube and 100 mg of hydrocortisone were injected intravenously. As a result, although arterial oxygen saturation on pulse oximetry increased to 95%, there was no change in PAP and the blood pressure dropped to 60/30 mmHg with an increased pulse rate of 120 beats/min (
Fig. 1). Absence of bleeding in the operative field was confirmed by the surgeon, but blood pressure was not restored by two injections of ephedrine 10 mg. Despite infusion of 15 Îźg/kg/min dobutamine and 0.2 Îźg/kg/min norepinephrine, the patient did not respond, and blood pressure remained at 65/40 mmHg. As bronchial spasm and hypotension that was unresponsive to medication were sustained, an anaphylactic reaction was suspected. When the patientâs skin lesions were evaluated after removing the surgical drape, erythematous rashes on both arms and severe swelling of the skin around the eyes were observed.
Fig. 1
Graphs of vital signs during surgery. At 25 minutes after the operation, pulse oxygen saturation dropped abruptly to 86% and blood pressure dropped to 60/30 mmHg with increased pulse rate of 120 beats/min. Epinephrine 0.5 mg was injected intravenously with hydration to treat the anaphylactic shock. As a result, blood pressure slowly increased to 100-120/50-70 mmHg, and pulse rate decreased to 80 beats/min.
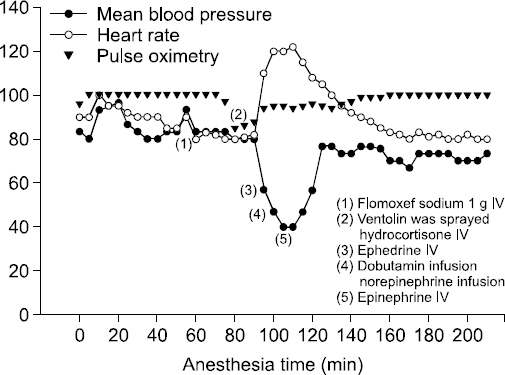
In order to treat the anaphylaxis, epinephrine 0.5 mg, pheniramine 40 mg, methylprednisolone 250 mg were injected intravenously with hydration. Blood pressure slowly increased to 100-120/50-70 mmHg, and pulse rate decreased to 80 beats/min. The wheezing sound disappeared, and the PAP fell to 16 cmH
2O. During the rest of the operation, vital status was stably maintained without medication other than intramuscular epinephrine 0.5 mg. At the completion of surgery, sugammadex 110 mg (Bridionâ˘, MSD, Netherlands) was given to reverse muscle relaxation. After spontaneous respiration and consciousness had recovered, the endotracheal tube was extubated and the patient was transferred to a recovery room. Except that systemic erythematous rash and swelling of the skin around the eyes could still be seen, there were no other abnormal findings. The skin symptoms resolved after 3 hours in the recovery room. Since the results of ABGA and chest radiography were normal and there were no further specific clinical presentations, the patient was transferred to a general ward. After 2 months, skin prick and intradermal tests were performed to establish the allergen. As a result, flomoxef sodium was identified as a cause of anaphylactic shock (
Table 1) and the patient was informed to avoid this agent.
Table 1
Results of Skin Tests to Agents
|
Drugs |
Skin prick test |
Intradermal test |
|
|
|
âConcentrations (mg/ml)â |
âResponseâ |
âConcentrations (mg/ml)â |
âResponseâ |
|
âHistamine (1 mg/ml) |
1 |
5 Ă 5 |
0.1 |
12 Ă 13 |
|
âSaline (control) |
- |
Negative |
- |
Negative |
|
âRocuronium (10 mg/ml) |
10 |
Negative |
0.05 |
3 Ă 3 |
|
âLidocaine (20 mg/ml) |
20 |
Negative |
2 |
2 Ă 3 |
|
âPropofol (10 mg/ml) |
2 |
Negative |
1 |
2 Ă 3 |
|
âFlomoxef sodium (250 mg/vial)â |
2 |
5 Ă 5 |
2 |
13 Ă 13 |
DISCUSSION
Because shock during the perioperative period is a problem that can directly threaten patient safety, its cause must be identified and it must be managed rapidly. In the present case, no abnormalities were found in the preoperative evaluations, and no change on electrocardiography or active bleeding during surgery was observed. Thus, the possibility of cardiogenic, neurogenic or hypovolemic shock was minimal, and anaphylaxis was suspected. The most common symptoms of anaphylaxis including symptoms of cardiovascular collapse such as hypotension, tachycardia or bradycardia, and arrhythmia, can be mistaken as effects of the anesthetic agents. In addition, mechanical ventilation after application of a muscle relaxant may mask important symptoms such as bronchial spasm, which can also make it difficult to diagnose. Moreover, even if anaphylaxis is identified, it is difficult to identify the cause due to the variety of agents to which the patient is exposed during the brief period of surgery, which can include multiple anesthetics, infusion solutions, latex, and antibiotics.
Of the anaphylactic reactions occurring under general anesthesia, 55-59% are caused by muscle relaxants, while latex (20-22%) and antibiotics (13-15%) are also major causes [
2,
6,
7]. The second generation cephalosporin, flomoxef sodium, was used in this case. Penicillins and cephalosporins that share the same chemical structure, β-lactam ring, are known as the major causes of antibiotic-related anaphylaxis which occupies up to 70-80% in proportion [
2,
6]. Moreover, anaphylactic reactions to antibiotics can even occur in patients catheterized with an antibiotic-coated central venous catheter [
8] and slow administration of vancomycin [
9].
The most important factors in diagnosing anaphylaxis are clinical presentations such as skin and mucosal involvement (pruritus, itchiness, and angioedema), respiratory symptoms (shortness of breath, bronchoconstriction, and stridor), and cardiovascular dysfunction (syncope, cardiovascular collapse, and hypotension) [
4]. Information on antigen exposure can point to the cause of anaphylaxis. In addition, anaphylaxis can be diagnosed by detecting an increase of serum tryptase, a neutral serine protease released by mast cells, within 1-2 hours after clinical symptoms and measuring its normalized serum level 24 hours later [
4,
7]. Lastly, skin tests such as the skin prick test and intradermal test are the gold standard for identifying the cause of immunoglobulin E mediated anaphylaxis [
6]. The skin test should be performed 4-6 weeks after anaphylaxis because of the high incidence of false negativity due to depletion of mast cells and specific immunoglobulin E antibodies [
4].
In the present case, the patient had undergone general anesthesia five times, and had a history of multiple uses of propofol, thiopental, opioids, vecuronium, rocuronium, latex, and other agents without hypersensitivity. Moreover, no anaphylactic reaction due to the multiple use of rocuronium was detected during this surgery. We were able to establish that this was the first time that flomoxef sodium was used in this patient by reviewing her medical records. We also verified that the anaphylactic reaction was due to flomoxef sodium, even though the preoperative intradermal test was negative. It is known that anaphylactic reactions can occur without previous exposure to a given medication due to structural similarity in drugs [
10]. In case of neuromuscular blocking agents, only 30-40% of patients have a history of previous exposure; in addition, cross-reactivity between antibiotics and neuromuscular blocking agents is common because of the structural similarity in these drugs [
3,
4,
11]. Furthermore, case reports of severe anaphylactic reactions with negative skin results [
12] and the finding that the sensitivity of the skin test for β-lactam antibiotics is only 50%, indicate that negative skin tests do not completely eliminate the risk of anaphylaxis [
13]. In addition, we could not exclude the possibility that the antibiotics were too much dilute to cause the anaphylaxis. Flomoxef sodium was diluted to 0.5 mg/ml for intradermal test before surgery. By comparison with the recommended concentration of 2 mg/ml, too much diluted concentration could be the reason why the preoperative intradermal test was negative. The tryptase test was not performed in this case because the anaphylactic skin lesions were obvious. However, anesthesiologists should be aware that the finding of elevated serum tryptase is an important indicator of anaphylaxis if skin reactions are unclear, and skin tests are negative [
14]. In addition, the possibility that anaphylactic reaction can occur 20-30 minutes after injection should not be overlooked [
15].
In conclusion, although antibiotics are known to be responsible for a significant fraction of anaphylactic reactions, the danger of anaphylaxis during general anesthesia employing antibiotics giving negative intradermal skin tests is not widely recognized. Negative results of intradermal skin tests do not completely eliminate the risk of anaphylaxis. Therefore, anesthesiologists should be prepared for the occurrence of anaphylaxis at any point in the perioperative period.





